Newsletter / Research Activity Report / Yuki Orikasa
RARA Newsletter Vol. 21 Capturing the Inside of a Battery during Operation and Transforming the Conventional Wisdom on Battery Research: Operando Measurement and the Potential of All-Solid-State Batteries—Professor Yuki Orikasa Rises to the Challenge
2026 / 01 / 06

2026 / 01 / 06
Observing Batteries During Charge and Discharge: The Innovation of Operando Measurement
This edition of the newsletter features an interview with Professor Yuki Orikasa (RARA Fellow; College of Life Sciences).

Prof. Orikasa’s research focuses on an innovative method known as operando measurement, which enables real-time observation of the very moment when batteries and fuel cells are in operation. Conventional battery research has largely relied on ex-post-analysis, in which batteries are disassembled and examined after use. Prof. Orikasa describes this approach to be “like looking at dried fish.” By contrast, operando measurement allows researchers to observe a battery while it is actually functioning, just like “watching a fish actually swimming.”
By utilizing SPring-8, one of the world’s largest synchrotron radiation facilities located in Hyogo Prefecture, and conducting computed tomography (CT) measurements with high-brilliance X-rays, changes inside a battery can be clearly visualized in three dimensions. This technique has revealed a series of dynamic phenomena occurring inside batteries—something that no other researcher has been able to observe before.
New possibilities for all-solid-state batteries: What the discovery of a “bridge” means
In October 2025, Prof. Orikasa’s research team published a groundbreaking discovery on all-solid-state batteries in the internationally recognized academic journal ACS Nano.
All-solid-state batteries are next-generation batteries that are expected to offer improved safety by replacing liquid electrolytes with solid ones. However, the silicon anode, which is the key to achieving higher capacity, presented a significant challenge. Silicon expands when it absorbs lithium ions and contracts when it releases them. Because solid electrolytes do not return to their original shape once deformed, it had been believed that gaps would form after the silicon contracted, interrupting the supply of lithium ions.
Yet, from the high-resolution X-ray CT images obtained using operando measurement, Prof. Orikasa’s research team discovered a surprising phenomenon. Even when the silicon particles contracted, parts of the contact with the solid electrolyte remained, forming something like a “bridge” and thereby preventing complete isolation. This was the first time in the world that researchers had made such a finding. The team further confirmed that, even after repeated charge–discharge cycles, degradation was significantly lower than in conventional batteries using liquid electrolytes.
This discovery has attracted attention for its potential to expand the use of silicon anodes in all-solid-state batteries substantially.
We spoke with Prof. Orikasa about the driving force behind his research, the course of development of the new method of operando measurement, moments of breakthrough achieved in collaboration with his students, and his views on the future of academia.
(The following is a summary of the conversation with Prof. Orikasa compiled by our writer.)
The evolution of batteries and expectations for all-solid-state batteries
Batteries, which are indispensable to our daily lives, have undergone a long history of evolution to their current form. Looking back on the history of battery research, it all began with the invention of the voltaic pile in 1800. In 1991, Sony became the first in the world to commercialize lithium-ion batteries, accelerating the widespread adoption of compact electronic devices such as mobile phones, laptop computers, and digital cameras.
Since then, batteries have expanded beyond mobile devices to applications including power tools, robots, electric vehicles, energy storage for renewable energy, and large-scale emergency power supplies and grid systems. As their range of applications has broadened, efforts to achieve larger capacity and higher energy density have continued to advance.
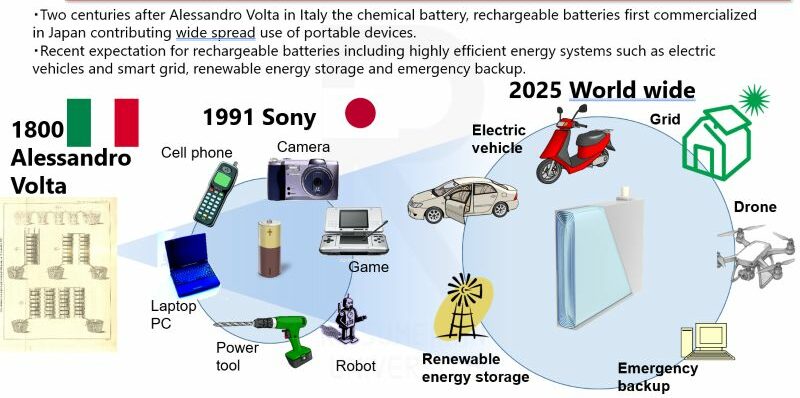
History and applications of batteries
First, let us look at the basic mechanism of a battery. Batteries repeatedly charge and discharge as lithium ions move back and forth between the positive and negative electrodes. The electrolyte facilitates this movement of ions. The lithium-ion batteries that are prevalent today use a liquid electrolyte. Lithium ions travel through this liquid electrolyte, allowing electrical energy to be extracted.
However, liquid electrolytes present certain issues. Because they use flammable organic solvents, there is a risk of ignition if the battery is damaged or becomes overheated. In recent years, reports of fires involving electric vehicles and smartphones have appeared in the news, and these incidents are related to the issues associated with liquid electrolytes.
Furthermore, as mentioned earlier, there is also the problem that the electrolyte concentration fluctuates while the battery is operating. When this happens, resistance may vary and uneven reactions may occur, affecting battery performance and safety.
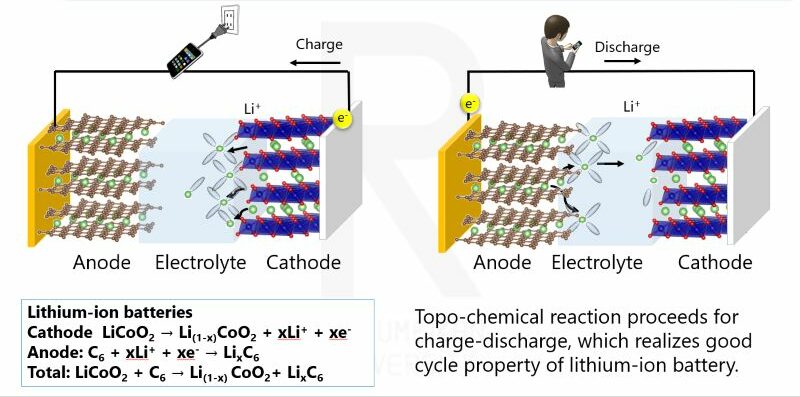
Lithium-ion battery reaction
This is why all-solid-state batteries have started to attract attention. All-solid-state batteries replace the liquid electrolyte with a solid one. Solid electrolytes are less flammable and eliminate concerns about leakage. They also offer the significant advantage that the electrolyte concentration does not fluctuate.
In fact, it has been known for more than ten years that the performance of lithium-ion batteries would eventually reach a plateau. The materials have changed little since they went on the market in 1991, and improvements in performance have largely depended on technologies that increase material density. However, the more densely materials are packed, the greater the risk of ignition becomes. To address these fundamental issues, the development of next-generation batteries is essential.
There are various candidates for next-generation batteries. Research and development have continued worldwide for more than a decade on lithium-metal batteries, sodium batteries, batteries using calcium or magnesium, batteries that operate by moving fluoride ions, and batteries that use air, to name a few.
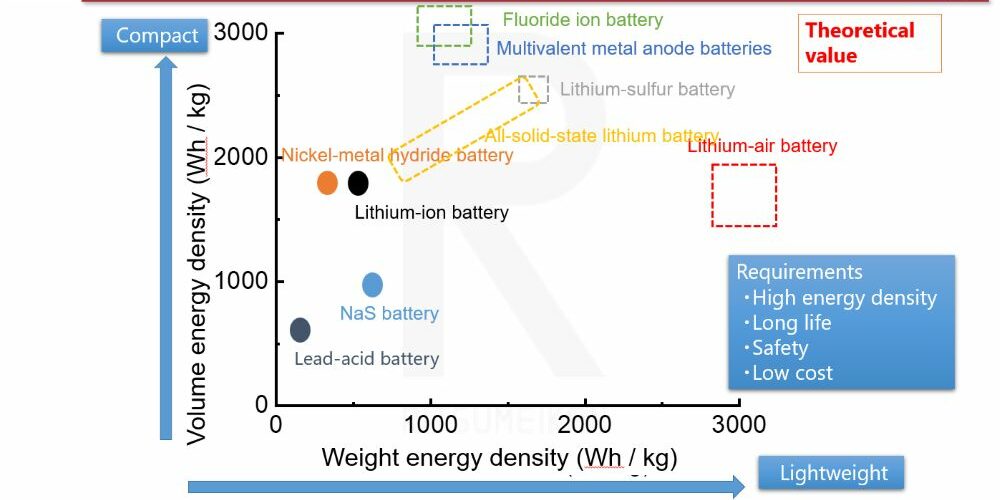
Energy density of various rechargeable batteries
Among these candidates, all-solid-state batteries have drawn particular attention because their materials are virtually the same as those of lithium-ion batteries, with the only difference being the electrolyte. This offers the advantage that the underlying chemical reactions can proceed more readily. While other battery systems involve new chemical reactions that often have slower reaction rates, all-solid-state batteries, despite potential limitations for further improvement, offer advantages such as reduced flammability and stable electrolyte concentration. Therefore, expectations are especially high for automotive applications.
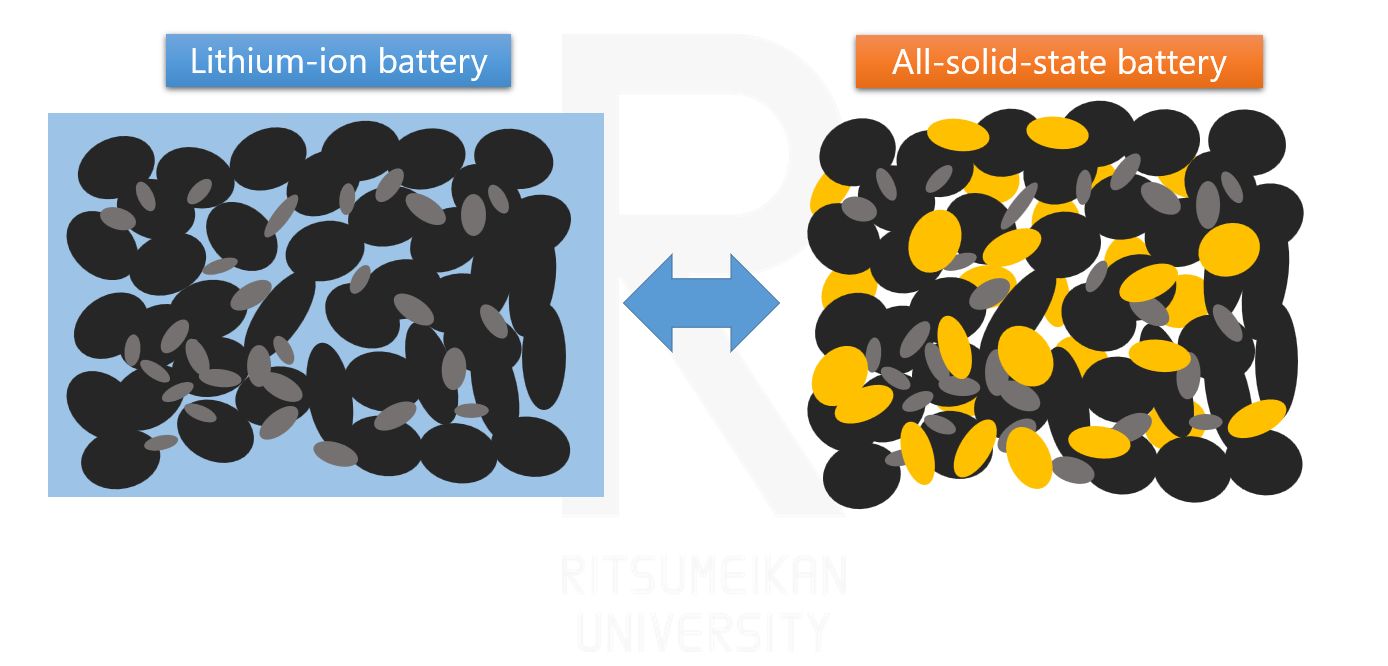
Differences between lithium-ion batteries and all-solid-state batteries
That being said, all-solid-state batteries are not yet an established technology, and it remains to be seen whether they can indeed be put into practical use. This is why operando measurement, which enables direct observation of batteries while they are actually operating, is a method whose importance is expected to increase.
“Like watching fish swim”: What is operando measurement?
In conventional battery research, the mainstream approach has been to disassemble and examine batteries after use. If we use the example of fish, this is like observing a dried specimen.
Operando measurement, by contrast, is a method that enables the observation of batteries while they are operating. To use the fish example again, this is akin to actually watching live fish while they are swimming. By observing a battery in operation, it becomes possible to understand things that were previously unknown and to apply those insights to battery design.
Dried fish do not move, so they are easy to observe, but living things move, making them more difficult to study. Observing a battery while it is operating is challenging, but it reveals phenomena that could not be seen before.

Ambitious research at SPring-8—Enabling the acquisition and analysis of vast amounts of data
We are conducting our experiments using SPring-8, the world-class synchrotron radiation facility located in Harima Science Garden City in Hyogo Prefecture.
There are only three synchrotron radiation facilities in the world today that are equivalent in size to SPring-8, and among them, SPring-8 produces the highest energy X-rays. In fact, some students approach me at the time of admission, saying, “I want to conduct experiments using SPring-8.”
Images that were out of focus in the laboratory can be taken very clearly at SPring-8. What’s more, we can use operando measurement to take images without the need to disassemble the battery. It is also possible to obtain data about ten times faster than in a laboratory, allowing an enormous amount of data to be collected.
However, because the experiments are conducted using equipment brought into the facility, preparation is quite demanding. Batteries are sensitive to moisture, so in the laboratory we use a glove box from which moisture has been removed, and this must be transported to the facility, which is 200 kilometers away. When we fabricate batteries and bring them to the site, there is no guarantee that they will operate properly, so we always travel there with a certain amount of anxiety.
If an experiment fails, we have to wait two to three months until the next opportunity opens up. It is therefore important to spend sufficient time on preparation and ensure that reliable data can be obtained. We carry out preparations together with our students, and we also work together with them on the day of the actual experiment. This takes a lot of time and is also mentally demanding.
There are also challenges in the analysis stage. Extracting important information from the enormous amount of data is difficult, and we spend a great deal of time on this task together with our students.
Recently, AI has become quite useful, so some tasks can now be handled by AI, thereby allowing the students to extract data from the results it produces.
For example, suppose that an image of a structure obtained through X-ray CT consists of three elements. Although the human eye can distinguish these elements, it is not feasible for a human to examine thousands of images one by one. Initially, humans create the data, but once the AI has been trained, it can carry out the rest of the work of identifying thousands of images. This allows us to conduct analyses using vast amounts of data.
Aiming to develop energy devices that can resolve environmental problems
The reason I entered battery research was that I wanted to work on energy devices that could help resolve environmental problems.
The background to this research lies in the expansion of energy use and the increasing severity of the environmental issues we face. Since the Industrial Revolution, energy-intensive development has led to a continual increase in CO₂ emissions, and the Intergovernmental Panel on Climate Change (IPCC) of the United Nations has concluded that “it is unequivocal that human influence has warmed the atmosphere, ocean and land.”
In response to this situation, an international shift toward carbon neutrality is underway. Through my research on energy devices such as batteries, fuel cells, and hydrogen technologies, I aim to contribute to this broader movement from a scientific perspective.
As for how I came to focus on research that looks inside batteries as they operate, it was really a matter of chance, as the area matched my own expertise. At a time when relatively few researchers were working on this topic, I have persevered in my research over many years, leading me to where I am today.
If we can understand what is happening inside batteries, we can get a better idea of the mechanisms behind phenomena such as ignition and degradation. Because this research is closely connected to everyday life, that connection serves as the driving force for my work.
As Japan’s market share continues to decline, all-solid-state batteries are becoming an option
Japan’s global share of consumer lithium-ion batteries accounted for 93% in 2000, shortly after their commercialization. Yet, this share declined to 72% in 2005 and to 43% in 2016, as competition in terms of both materials and cost intensified. It has been said that unless we can identify the next generation of batteries, Japan’s battery industry will fall into decline.
These changes in industrial structure have also reinforced the need for next-generation batteries. While awareness of the importance of next-generation batteries has spread, there is still no clear answer as to what form they will take.
The discovery of a “bridge”: New possibilities for all-solid-state batteries
The study we published in ACS Nano in October 2025 was carried out primarily by graduate students who graduated this past March.
Using operando measurement, our team observed an all-solid-state battery at high resolution and conducted a thorough analysis. The graduate students compiled the work on their own with the cooperation of myself and the other faculty members on the team.
Silicon is an attractive material because it can absorb a large amount of lithium ions, but it expands during charging and contracts during discharging. In lithium-ion batteries, the holes that form when the silicon contracts allow the electrolyte to react, leading them to degradation. For this reason, it is considered difficult to use silicon alone.
It was initially thought that the same problem would occur in all-solid-state batteries. Because solid electrolytes do not return to their original shape once deformed, gaps form once the silicon contracts, so we expected the supply of lithium ions to stop. A liquid electrolyte can move in and fill these gaps, but a solid one does not move, so the supply would stop. In theory, this was what we all assumed.
Two new findings emerged from this study, however. One was that silicon does not become completely isolated; something like a “bridge” with the solid electrolyte remains in small quantity.
When silicon expands and contracts, gaps form, but even after repeated cycles, part of the solid electrolyte adheres to the electrode surface and maintains contact. Because these bridges remain, lithium ions can continue to move.
We had begun to observe signs of this about three years ago. A student at the time identified the presence of these bridges, obtained clear images, observed intermediate states, and repeated the experiments many times to confirm their reproducibility.
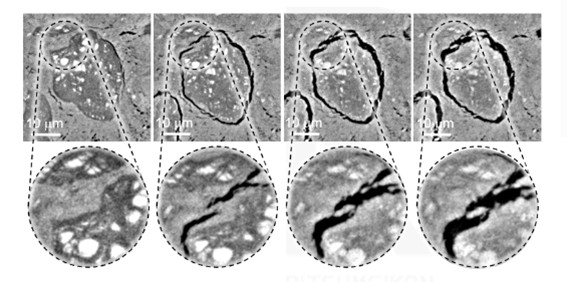
A bridge formed between the solid electrolyte and the silicon anode as captured by operando measurement
Seeing the actual images with X-ray CT is important. Many studies rely on a method called spectroscopy and discuss results using only line graphs, but with actual images, it is much easier to directly understand what is happening inside the battery, which also makes it easier to explain to a general audience. With an image in front of you, you can clearly see that the silicon anode and the solid electrolyte are indeed connected. We had never seen such a crisp image of the inside of an all-solid-state battery before; only much blurrier images were available. This is because scanning precision was not as good as it is now.
The other discovery was that even after repeated charge–discharge cycles, degradation is suppressed compared with liquid electrolytes, meaning that, in this respect, all-solid-state batteries perform better. The shape remains clearly preserved, no matter how many times the process is repeated, and the bridge portions do not change much. Cracks do appear, but the bridges always form in the same places and move quite dynamically.
With this finding, we were able to move beyond the assumption that silicon could not be used in all-solid-state batteries. There are still issues, but what had been regarded as a major drawback turned out not to be one.
That is how development works. Progress does not usually come in a single dramatic leap after one problem is solved; rather, it advances through the gradual accumulation of improvements, refinements, and discoveries. This was also the case with lithium-ion batteries.
This time, the fact that we were able to make a discovery through operando measurement is significant. With operando measurement, it is possible to continue observing changes over time in the same sample. With ex-post-analysis, you can only line up different states and infer what happened, making the results ambiguous.
In fact, at first, we observed holes forming in the silicon; we thought this would demonstrate that silicon was not suitable after all. However, when we looked more closely, we realized that the battery continued to function, and focusing on that aspect led to our discovery.
Research is built on failure, so you must welcome the unexpected
Research generally involves many failures, and things rarely proceed exactly as planned. To increase the chances of success, I conduct thought experiments. Before beginning an experiment, I think carefully about what kind of results I would like to obtain. I also consider what might go wrong and put those possibilities into some semblance of order before moving forward with the research. This process often leads to discoveries.
The seeds of future discoveries are often found in dialogue. Through interviews like this one or conversations with people in industry, I gain different perspectives and link them to my own research, which helps reveal new points for investigation.
Sometimes this is done intentionally, but at other times it happens naturally. I place great importance on that kind of serendipity. I do not believe that I can accomplish this work alone. I hope to make unexpected discoveries that originate from ideas I had not previously considered.
In fact, this discovery began when a student from two graduating classes ago said they wanted to pursue this line of research. We had been proceeding with the idea that we would obtain data showing that silicon would not work, but instead we discovered something new.
Basically, I only lecture students on the methodology, and then I let them carry out the experiments as they see fit. When they have difficulty extracting information, I look at the data and tell them we need to consider this or that, but quite often the students end up identifying new phenomena on their own accord. I try to create as many opportunities for this to happen as possible.
The happy problem of being in an environment that allows me to concentrate on research—My reflections as a RARA Fellow
By making use of the RARA Fellowship Program, the amount of time I can devote to research has increased considerably. I now have more time for thinking and engaging in discussions, especially with students. Because we are conducting research where even we ourselves do not know what results will emerge, I need to work hard together with my students. I am grateful that the program has been designed in a way that affords me more of this kind of time.
When students say, “I want to try this analysis,” or “I don’t understand this part,” we can sit down together, think it through, and move the research forward by discussing possible approaches. I am very grateful for this.
Thanks to RARA, I am now able to pursue a variety of research interests, and I am receiving all kinds of requests. I am currently involved in national-level projects in areas such as lithium-ion batteries, all-solid-state batteries, fuel cells, and hydrogen technologies. Things have become quite busy, and the number of commissioned research projects is increasing. It’s really a happy problem. I intend to continue making full use of this environment going forward.
A desire to contribute to society through energy device research
My research does not directly lead to carbon neutrality. Our work becomes relevant only after a material is discovered and we start to question why something happens. Our role is to provide support when new technologies emerge.
By understanding what is happening while devices are operating, we study whether they can function properly. This allows us to determine whether new materials are performing as expected. If they are not, we can change direction more quickly, which helps accelerate the pace of research and development.
What we are currently trying to do is work together with industry to steadily advance both materials development and evaluation and analysis. As an academic, I would like to contribute by considering what is truly useful for development.
I would like to serve as a bridge between practical use and academia in the areas of energy, batteries, and fuel cells. My specialty is electrochemistry, so I hope to utilize and further develop my expertise and measurement techniques as I continue to pursue interesting research well into the future.
Restoring the appeal of academia as a place to enjoy intellectual debate
In Japan today, fewer students are pursuing doctoral studies in the sciences, and the number of people choosing to remain in academia is declining. Concerned that the next generation of researchers may disappear, I would like to change this situation. From a global perspective, Japan is somewhat unusual in that universities tend to function almost like preparatory schools for employment, and students seem to want to secure a job as early as possible.
The word “philosophy” originates from the ancient Greek words philo, meaning “love,” and sophia, meaning “wisdom,” so it literally means “love of wisdom.” Similarly, the word “school” in English derives from the Greek scholē, which originally referred to “leisure” or “free time.” It reflects a culture in which using one’s free time for reflection and discussion was considered valuable.
The Doctor of Philosophy (PhD) degree is positioned as a “passport to world-class academia” linked to these philosophical traditions. The true starting point of academia lies in the joy of intellectual inquiry and debate driven by pure curiosity. I want students to experience the excitement of discovery, which is something that cannot be measured simply by whether it is “useful.”
In ancient Greece, academia and universities were places where people gathered to enjoy discussion. I would like to make the university a place where research and intellectual debate can truly be enjoyed. It does not have to be about whether something will be useful in the future. For example, for my students, it would be enough to simply think, “Discovering that bridge has been remarkable.” I hope more students can experience that sense of discovery and the joy that comes with it.
There is also much discussion today about whether human work will be eliminated in the age of AI. In that context, I believe it will become increasingly important to simply enjoy academia itself, as the community in which we learn, conduct research, and make discoveries. Academia should be a place where researchers can concentrate on their work without worrying too much about whether it is immediately useful or not. My own research happens to have close ties to industry, but I believe university is a place where such concerns do not have to dictate what we do.
Recently, many symposia have become rather formal, with little genuine discussion among participants. Even when we encourage students to gather and engage in serious debate, it does not always happen easily. I would like to explore new formats for symposia and gradually restore an environment where such open intellectual exchange can naturally flourish again.




